Johnson & Johnson pauses Covid-19 vaccine trial
Washington, Oct 13: US pharmaceutical giant Johnson & Johnson announced that it has temporarily paused its Covid-19 vaccine trial after a participant became unwell."We have temporarily paused further dosing in all our Covid-19 vaccine candidate clinical trials, including the Phase 3 ENSEMBLE trial, due to an unexplained illness in a study participant," Xinhua news agency quoted the company as saying in a statement on Monday.The patient's condition was being reviewed and evaluated by the ENSEMBLE independent Data Safety Monitoring Board as well as internal clinical and safety physicians, it added."Based on our strong commitment to safety, all clinical studies conducted by the Janssen Pharmaceutical Companies of Johnson & Johnson have pre-specified guidelines. These ensure our studies may be paused if an unexpected serious adverse event (SAE) that might be related to a vaccine or study drug is reported, so there can be a careful review of all of the medical information before deciding whether to restart the study," the company statement further said."We must respect this participant's privacy. We're also learning more about this participant's illness, and it's important to have all the facts before we share additional information."Serious adverse events are not uncommon in clinical trials, and the number of serious adverse events can reasonably be expected to increase in trials involving large numbers of participants. Further, as many trials are placebo-controlled, it is not always immediately apparent whether a participant received a study treatment or a placebo," it addedThe vaccine is being developed by the company's Janssen arm, CNN said in a news report.The company however, did not mention what the illness was.Trials are stopped when they pop up while doctors check to see if the illness can be linked to the vaccine or is a coincidence.Johnson's Phase 3 trial started in September and it id one of six coronavirus vaccines being tested in the US, and one of four in the most advanced, Phase 3 stage.This is the second Phase 3 coronavirus vaccine trial to be paused in the US.AstraZeneca's vaccine trial was paused last month because of a neurological complication in a volunteer in the UK.While the trial resumed in the UK and in other countries, it remains halted in the US while it is being probed by the Food and Drug Administration investigates. (IANS)
Latest News

Back on court, Nadal outplays Blanch in Madrid...

Students of Rama Devi Women’s University prote...

IPL 2024: Do chase better, Vettori advises SRH...

Two IPLs simultaneously in India!

PM Narendra Modi blames Mamata government for...

Young girl dies in elephant attack in Odisha’s...
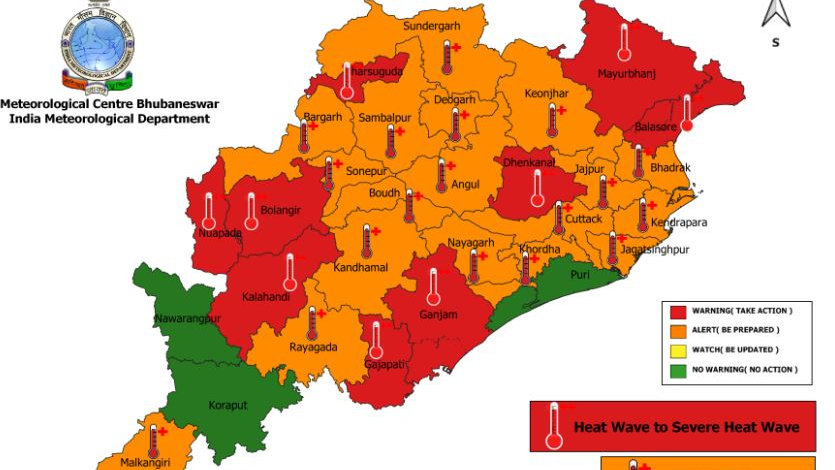
Odisha continues to grapple with intense heatw...
Copyright © 2024 - Summa Real Media Private Limited. All Rights Reserved.